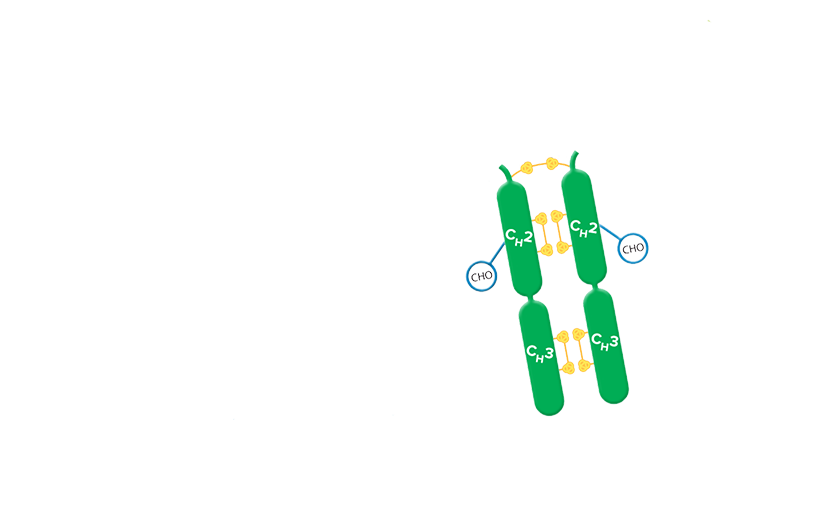
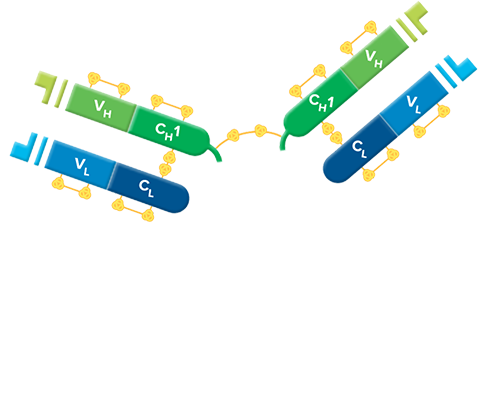
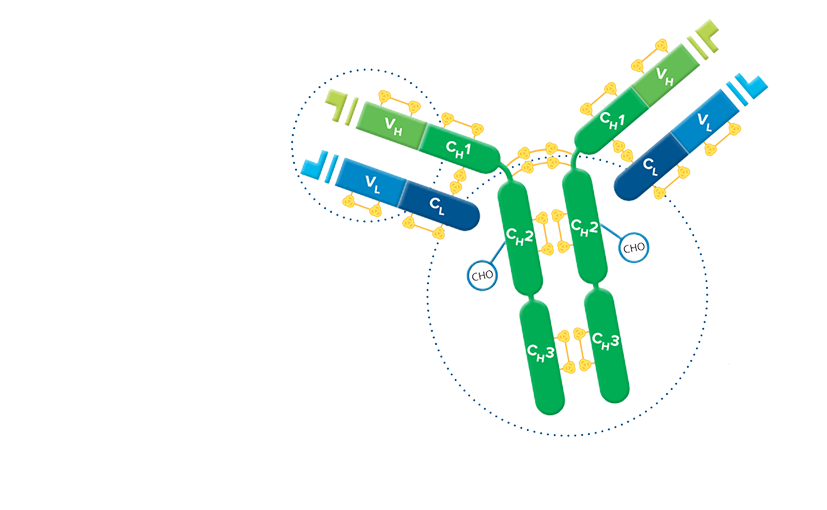
F(ab')2 fragment antibodies are generated by pepsin digestion of whole IgG antibodies to remove most of the Fc fragment while leaving some of the hinge region intact. F(ab')2 fragments have two antigen-binding (Fab) regions linked by disulfide bonds, making them divalent. The average molecular weight is approximately 110 kDa. They are used in specific applications, such as avoiding recognition of secondary antibodies by Fc receptors on live cells, or to Protein A or Protein G

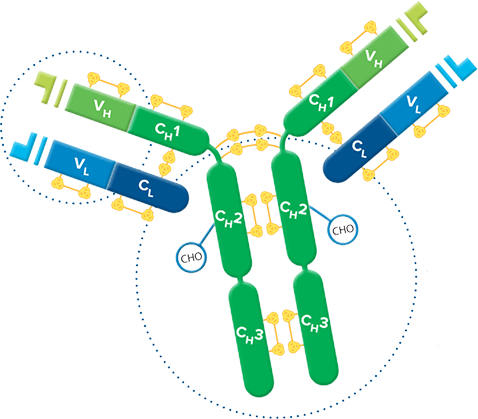
Binding of primary antibodies to Fc receptors also may occur if they are whole IgG antibodies, creating background regardless of the secondary antibody format. To block whole IgG primary and secondary antibodies from binding to Fc receptors, incubate cells in buffer containing 5% normal serum from the host species of the labeled secondary antibody. To prevent capping, endocytosis, and regeneration of Fc receptors on living cells, incubate at 4°C in buffer containing 5% normal serum with sodium azide added to inhibit metabolism.
We also have a limited inventory of DyLight™ 488 / 549 / 594 / 647, Cy™2, Cy™5, and Texas Red® conjugated secondary antibodies.