
"The anti-human polyclonal capture reagents are some of the most robust reagents I have used in my biophysical and biochemical studies so far. Overall, the anti-Fc reagents regardless of the host or capturing species provides a robust capturing ability with minimum cross-reactivity to other species." Full review.
Rupesh Nanjunda,Rating: 5.0

"I have used a wide variety of secondaries and Jackson ImmunoResearch has consistently been the best. The fluorophores are bright and stable and their selective (x reactivity removed) secondaries have always shown species specificity in multiple labeling."
Janet Duerr, Ohio UniversityRating: 5.0
Serological tests are key assays for many health issues, including solid organ transplantation screening, crossmatching, disease diagnosis, monitoring and management. These assays play a crucial role in clinical decision-making. Immunoassays are among the most powerful and sensitive serological tests available, based on highly specific binding between antibodies and antigens, enabling qualitative and quantitative detection of analytes even at low concentrations. There are a variety of immunoassays available and the detection process varies from technique to technique. The most common of which are illustrated below showing a sandwich (Fig. 1A) and direct bind (Fig. 1B), respectively. When choosing an Anti-Human Secondary Antibody for use in sandwich format assays (Fig. 1A), it is important to consider the species in which the primary antibody was generated and to use a secondary antibody that has minimal cross-reactivity to that species.

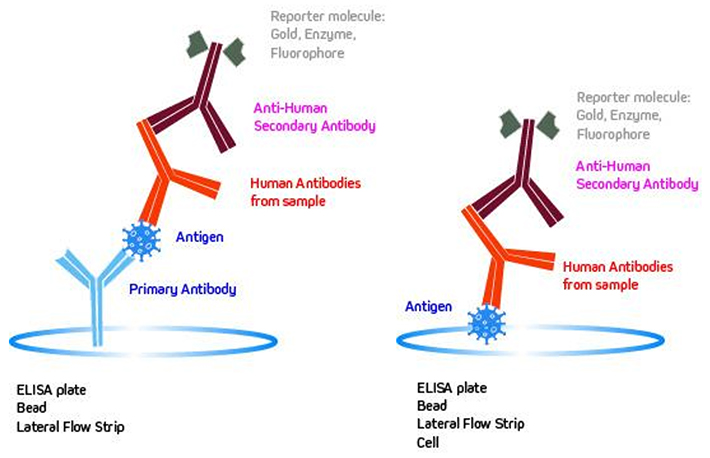
Immunoassay techniques each have advantages and limitations. For example, rapid test assays like lateral flow provide qualitative results within minutes, allowing for point of care analysis, where as an ELISA can take hours to perform but provides quantitative information.

The benefits of lateral flow assays are highlighted by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. Considering the fast spread and mortality rates of the disease, the FDA has taken action to allow states to launch new SARS-CoV-2 tests without FDA approval. This amendment allows the use of rapid test kits, which qualitatively detect IgG and IgM patient-generated antibodies to SARS-CoV-2, expanding the SARS-CoV-2 diagnostic capacity, with the aim of reducing the spread of the virus. Although there are clear advantages to lateral flow tests, these immunoassays don’t offer quantitative data, which is a key feature for some clinical tests.
Another critical application for which Anti-Human Secondary Antibodies are utilized is the transplant crossmatch assay. Flow cytometry crossmatch is an essential immunoassay when determining the suitability of a transplant patient for a donor organ. In these assays, the presence of donor-specific crossmatch antibodies in the organ recipient to human leukocyte antigens on the donor’s white blood cells, or on bead conjugates, is determined. The information provides the histocompatibility of the donor and the recipient and clinicians can then assess the suitability for organ transplantation.

| Immunoassay | Detection Process | Immunoglobulin Isotypes |
|---|---|---|
| Rapid test/ lateral flow | Sample is loaded onto a test membrane and travels by capillary action. Analytes, including hyperimmune antibodies, present in the sample bind to color-coded Anti-Human detection antibodies as they travel. The complexes bind to either an immobilized sandwiching antibody (Fig. 1A) or an antigen in the form of a test line (Fig. 1B). A colored line provides a qualitative positive result. A common method of color-coding reporter antibodies is through conjugation to gold nanoparticles. A control line is generally also included that indicates the assay is functioning properly. |
IgG, IgM |
| Flow cytometry | A serum sample is added to antigen-bearing cells or coated beads. A secondary antibody conjugated with a fluorescent probe is added and binds to any human antibody captured on the cell or beads (Fig. 1B). The fluorescence can then be analyzed by the flow cytometer to give quantitative data. | IgG, IgM, IgA |
| Flow Injection Analysis (FIA) | Similar to flow cytometry, the sample is injected into a column containing immobilized antigen, to which the antibodies of interest in the sample binds (Fig. 1B). A secondary antibody is added and binds to any human antibody captured on the column. This secondary antibody can either be conjugated to a fluorescent probe or an enzyme for colorimetric or fluorescent detection. | IgG, IgM, IgA |
| ELISA | A primary antibody is immobilized on a solid surface, typically a microtiter plate, which subsequently captures the antigen (Fig. 1A) or the antigen is coated directly onto the plate (Fig. 1B). Human antibodies from hyperimmune serum bind to the antigen. An enzyme-conjugated Anti-Human Secondary Antibody is added which binds to the human antibody from the sample. Finally, an enzyme substrate is added, which results in a colorimetric readout proportional to the analyte concentration. | IgG, IgM, IgA |
| IPCR | This process is similar to ELISA, but the secondary antibody is conjugated to an oligonucleotide. DNA amplification and detection are then performed by real-time PCR. | IgG, IgM, IgA |
While the function and purpose of immunoassays differ, high-quality reagents are essential when manufacturing any serological kit to produce accurate and reliable data that clinicians and patients can trust. The quality and characteristics of Anti-Human Secondary Antibodies are crucial considerations when developing an immunoassay. Secondary antibodies are preferred as they bind multiple epitopes on one primary antibody, which enhances the signal for higher sensitivity and selectivity.
Table 2 describes the many aspects to consider when selecting the most suitable secondary antibodies for an immunoassay.
Jackson ImmunoResearch Laboratories, Inc. specializes in providing secondary antibodies for immunological applications. Their wide range of products enables customers to select the most suitable secondary antibody for the development of their serological tests.
All Anti-Human Secondary Antibodies are manufactured to ISO 9001:2015 certification, providing quality, specificity and reliability of their products and these secondary antibodies can be purchased in standard and bulk quantities, which can be shipped worldwide.
| Consideration | Jackson ImmunoResearch Secondary Antibodies | |
|---|---|---|
| Host Species | Serological tests for human samples should use Anti-Human Secondary Antibodies to minimize non-specific binding, which can lead to high background signals and false positives. | Anti-Human Secondary Antibodies are developed in a range of host species including alpaca, donkey, goat, mouse and rabbit. |
| Product type | Secondary antibodies are available as whole immunoglobulin (Ig)G, F(ab’)2 and Fab fragments. Whole IgG antibodies are often sufficient, but F(ab’)2 and Fab fragments are useful to avoid binding to live cells with Fc receptors. | Secondary antibodies are available as whole IgG and F(ab’)2 and Fab fragment formats. |
| Specificity | Depending on the specificity of the immunoassay required, secondary antibodies can be made to be specific to the whole Ig, Fc or F(ab’)2 domain. | Anti-Human Secondary Antibodies offer specificity for whole Ig, Fc and F(ab’)2 domains. |
| Isotype | Most immunoassays test for IgG isotypes, which constitute ~75% of the total serum Ig and are produced as part of the secondary immune response. However, IgM and IgA can be used, as these function in the primary immune response and protect mucus membranes, respectively. It is important that the secondary antibody has specificity for the isotype of the primary antibodies. | Products are available with specificities to IgG, IgM, IgA, IgG+IgM and IgG+IgM+IgA. |
| Affinity purification and cross-adsorption | Due to the high structure conservation in Ig domains, it is recommended to use class or species specific secondary antibodies that have been affinity purified and cross-adsorbed to reduce the possibility of cross-reaction. | Immunoaffinity chromatography is used to isolate affinity-purified antibodies. Antibodies can be purchased which have been cross-absorbed against species and these details are provided in the parentheses of the product description with the term “min X” followed by abbreviations for the relevant species. |
| Conjugate | Secondary antibodies are often conjugated to a reporter molecule e.g. an enzyme, fluorescent probe, or colored particle. The detection system of the immunoassay will determine the type of conjugate. | Anti-Human Secondary Antibodies are available unconjugated or conjugated to a range of reporter types, including biotin, alkaline phosphatase, horseradish peroxidase, fluorophores and colloidal gold nanoparticles. |