This article introduces key concepts underpinning cancer research, including the hallmarks of cancer and major historical milestones. It explores how antibody-based immunotechniques, particularly Western blotting, enable the investigation of protein-level changes that drive disease, providing a framework for understanding cancer biology and guiding experimental approaches in modern oncology research.

Immunotechniques for Cancer Research
Introduction and the Hallmarks of Cancer
The International Agency for Research on Cancer (IARC), which forms part of the World Health Organization (WHO), estimates that there were 20 million new cancer cases in 2022 – a number that is predicted to surpass 35 million in 2050. The rapidly increasing global cancer burden, driven by population aging and growth, underscores the critical importance of scientific research to understand disease pathogenesis and develop effective treatments.
Cancer research is challenging since cancer is the result of a unique combination of genetic and epigenetic changes. These are induced by aging, as evidenced by shared alterations in aged cells and cancer cells (Zabransky et al., 2022). Additionally, genetic changes may be caused by environmental exposures such as mutagenic chemicals and ultraviolet (UV) light, while epigenetic changes have been linked to factors including chronic inflammation (Maiuri et al., 2016), obesity (Long et al., 2020), stress (Abate et al., 2020), and perturbation to the microbiota–host relationship (Woo & Alenghat, 2022). Determining the impact of these events is essential to improving patient outcomes.
Key Milestones in Cancer Research
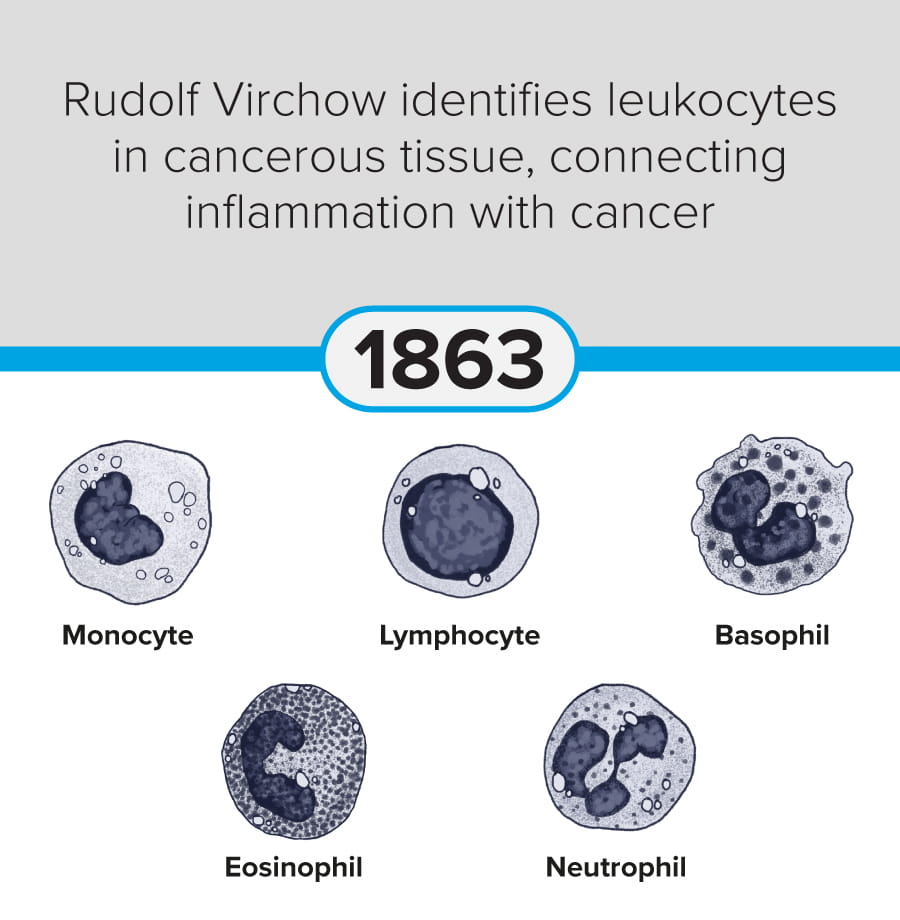
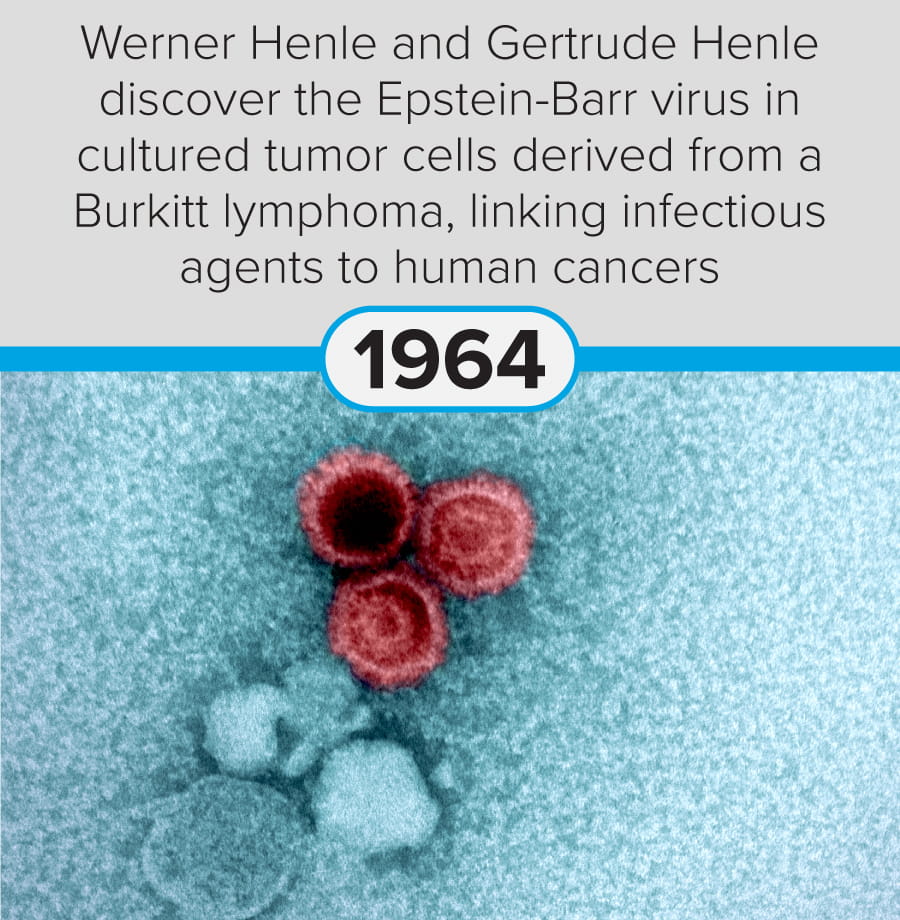
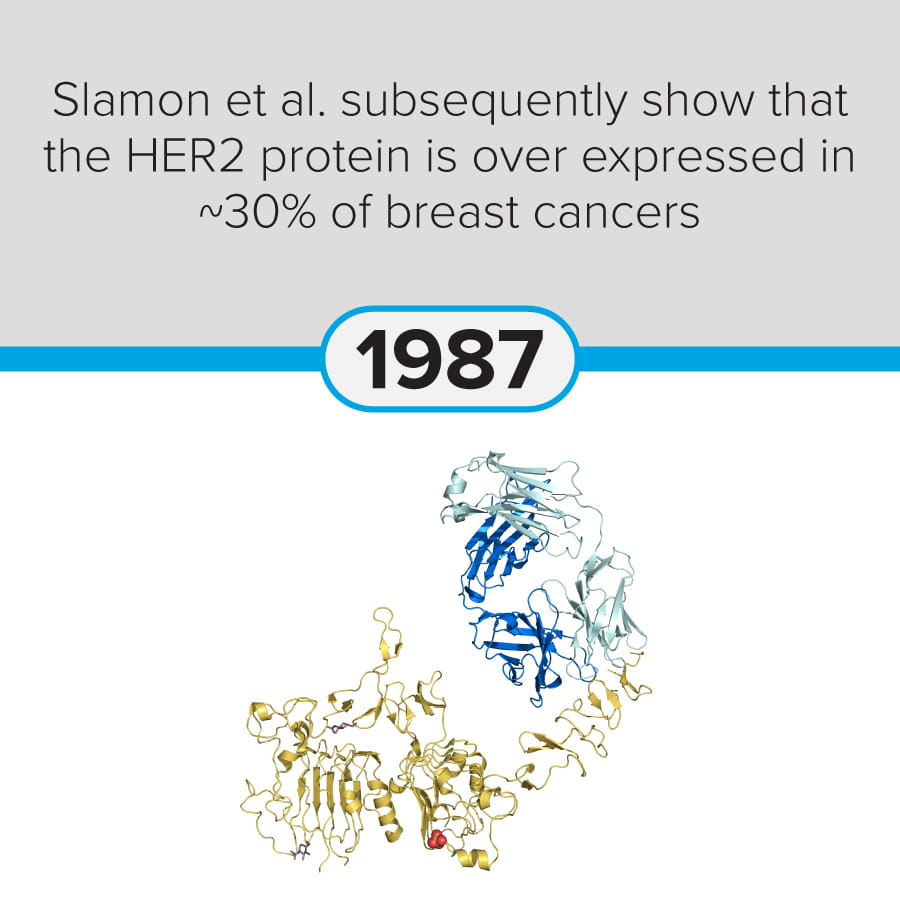
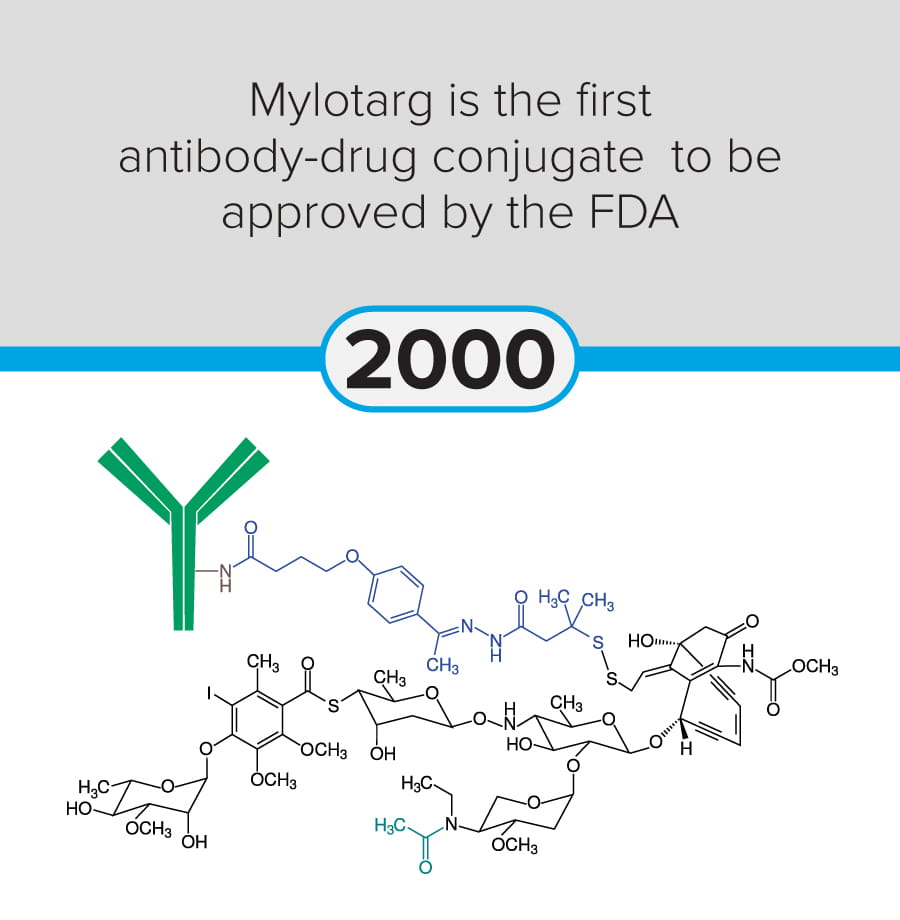
The first steps in cancer research date back to at least 1775, when Percivall Pott linked occupational exposure to soot with testicular cancer (chimney sweeps’ cancer). Since then, modern-day breakthroughs include significant advances in mRNA vaccine technology (Yaremenko et al., 2025) and the development of a novel plasma test (Budnik et al., 2023) to identify early-stage cancers.
The following discoveries represent key milestones from the last 250 years:
These milestones demonstrate the progression of cancer research from early pathological observation to modern molecular, immunological, and genetic and genomic therapeutic approaches.
The Hallmarks of Cancer
The hallmarks of cancer, first proposed by Douglas Hanahan and Robert Weinberg in 2000, are defined as six essential alterations in cell physiology that collectively dictate malignant growth:
- Self-sufficiency in growth signals
- Insensitivity to growth-inhibitory signals
- Evasion of programmed cell death (apoptosis)
- Limitless replicative potential
- Sustained angiogenesis
- Tissue invasion and metastasis
These disruptions to normal processes are enabled by genome instability and mutation, often in key regulators of cellular function. For example, a mutation in the epidermal growth factor receptor (EGFR) or the tumor suppressor gene p53 can lead to uncontrolled growth.
In 2011, Hanahan and Weinberg suggested two further hallmarks, the reprogramming of energy metabolism and evading immune destruction, and a second enabling characteristic, tumor-promoting inflammation. In 2022, four additional prospective hallmarks and enabling characteristics were recommended shown in Figure 1 (Hanahan, 2022), reflecting advances in the understanding of tumor biology.
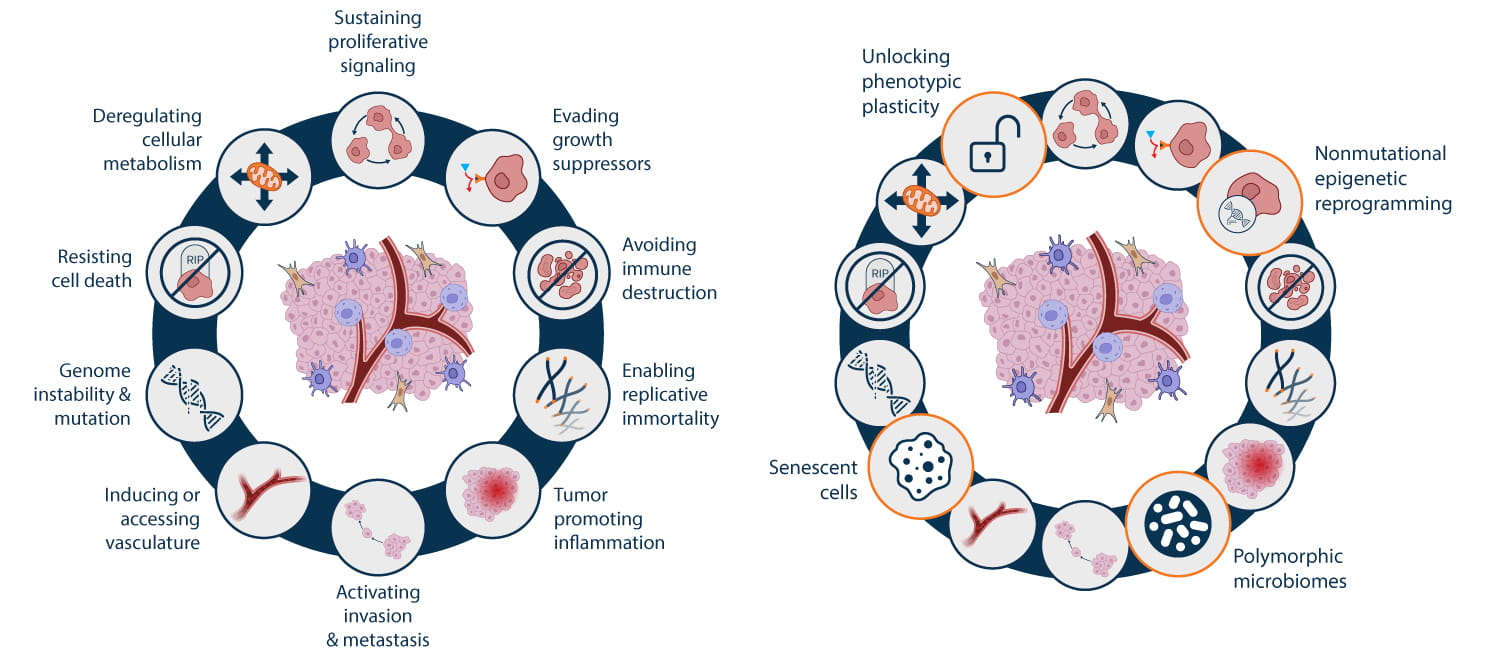
This ongoing refinement of the hallmarks of cancer demonstrates the need for dynamic investigation based on detailed experimental design. To accomplish this, cancer research has become segmented into more focused areas, many of which overlap with broader fields such as inflammation, immunity, and epigenetics. By interrogating distinct aspects of cancer pathogenesis using specialized techniques, researchers can better dissect intricate experimental questions. However, maintaining a holistic overview of cancer remains fundamental to improving clinical outcomes.
From Concept to Investigation
The hallmarks framework provides a structured way to interpret the biological consequences of genetic and epigenetic alterations. Each hallmark corresponds to molecular and cellular processes that can be examined experimentally.
Cancer is characterized not only by changes at the DNA and RNA levels, but also by alterations in protein expression, modification, localization, and interaction. Proteins serve as the functional mediators of cell growth, survival, differentiation, and migration. Consequently, protein-level analysis remains central to understanding oncogenesis.
Antibody-based immunotechniques allow researchers to detect specific proteins within complex biological samples, assess relative expression levels, and examine post-translational modifications that regulate protein function. These approaches enable insight into cancer-associated processes including signaling pathway activation, immune evasion, angiogenesis, and metastasis.
The hallmarks of cancer therefore provide both a conceptual foundation and a practical framework for experimental design, guiding investigations across diverse areas of oncology research.
Technique Focus: Western Blotting in Cancer Research
Cancer is a complex disease characterized by genetic mutations and widespread dysregulation of cellular signaling pathways. While changes at the DNA and RNA levels contribute to oncogenesis, it is the resulting alterations in protein expression, modification, localization, and interaction that ultimately drive malignant behavior. Proteins serve as the functional mediators of cell growth, survival, differentiation, and migration, making protein-level analysis essential for understanding cancer biology. Western blotting enables researchers to detect specific proteins within complex biological samples, assess relative expression levels, and examine post-translational modifications that regulate protein function, enabling insight into cancer-associated processes.
A common application is assessing target engagement following therapy. For example, Xia et al. (2002) used Western blotting to show reduced HER2 phosphorylation and downstream signaling in breast cancer cells treated with the EGFR/HER2 inhibitor lapatinib, confirming pathway inhibition. Western blotting also complements discovery-based approaches by validating key signaling events, as demonstrated by Rikova et al. (2007), who confirmed oncogenic kinase phosphorylation in lung cancer samples identified by phosphoproteomics. In studies of drug resistance, Western blotting remains essential for revealing adaptive signaling changes. Chandarlapaty et al. (2011) used this approach to demonstrate compensatory activation of the MAPK/ERK pathway in PI3K inhibitor–resistant breast cancer models, highlighting the importance of protein-level analysis in understanding therapeutic failure.
Principles of Western Blotting
Western blotting is an antibody-based technique that combines protein separation with specific immunodetection. The process begins with the extraction of total protein from cells or tissues, typically using detergent-based lysis buffers that preserve protein integrity. Proteins are then denatured and separated by size using sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE). This step allows complex protein mixtures to be resolved into discrete bands based on molecular weight.
Following electrophoresis, the separated proteins are transferred to a membrane, typically nitrocellulose or polyvinylidene difluoride (PVDF). The membrane is then blocked to prevent nonspecific antibody binding and incubated with a primary antibody that specifically recognizes the protein of interest. Detection is achieved using a secondary antibody conjugated to an enzyme or fluorophore, enabling visualization of the target protein as a distinct band.
A key strength of Western blotting is its ability to distinguish between different forms of the same protein. By using antibodies that recognize specific post-translational modifications, such as phosphorylation or cleavage, researchers can assess protein activation states rather than simply total abundance. This capability is particularly important in cancer research, in which aberrant signaling often arises from changes in protein activity rather than expression alone.
Product Focus: Fluorophore-Conjugated Secondary Antibodies and Multiplex Western Blotting in Cancer Research
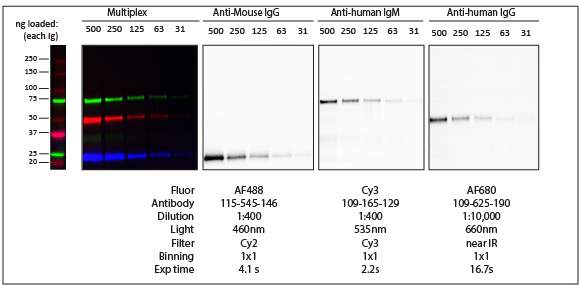
While HRP-conjugated secondary antibodies remain a standard option for Western blot detection, fluorophore-conjugated secondary antibodies add benefits such as suitability for quantitative and multiplexed analyses. Fluorescent detection offers a broad linear dynamic range and stable signal, enabling more accurate measurement of protein expression and post-translational modifications in complex cancer samples.
Jackson ImmunoResearch (JIR) fluorophore-conjugated secondary antibodies are available with spectrally distinct dyes, allowing multiple targets to be detected simultaneously on a single blot. This multiplex capability is particularly valuable in cancer research, in which coordinated changes across signaling pathways are common. For example, tumor markers in their unphosphorylated and phosphorylated states, with a loading control, can be analyzed in parallel without stripping and re-probing, reducing variability and conserving limited samples.
| Fluorophore | Excitation Peak (nm) | Emission Peak (nm) |
|---|---|---|
| DyLight™ 405 | 400 | 421 |
| Alexa Fluor® 488 | 493 | 519 |
| Indocarbocyanine, Cy™3 | 550 | 570 |
| Alexa Fluor® 555 | 552 | 572 |
| Alexa Fluor® 568 | 577 | 602 |
| Alexa Fluor® 594 | 591 | 614 |
| Alexa Fluor® 647 | 651 | 667 |
| Indodicarbocyanine, Cy™5 | 650 | 670 |
| Alexa Fluor® 680 | 684 | 702 |
| Alexa Fluor® 790 | 792 | 803 |
In the next article, we move from conceptual foundations to one of the most influential areas of contemporary oncology: the tumor microenvironment, a dynamic ecosystem that shapes tumor progression and therapeutic response.
References
Abate M, Citro M, Caputo M, et al. Psychological stress and cancer: New evidence of an increasingly strong link. Transl Med UniSa. 2020;23:53–57. doi:10.37825/2239-9747.1010
Anderson NM, Simon MC. The tumor microenvironment. Curr Biol. 2020;30(16):R921–R925. doi:10.1016/j.cub.2020.06.081
Asmamaw Dejenie T, Tiruneh G/Medhin M, Dessie Terefe G, et al. Current updates on generations, approvals, and clinical trials of CAR T-cell therapy. Hum Vaccin Immunother. 2022;18(6):2114254. doi:10.1080/21645515.2022.2114254
Boczkowski D, Nair SK, Snyder D, Gilboa E. Dendritic cells pulsed with RNA are potent antigen-presenting cells in vitro and in vivo. J Exp Med. 1996;184(2):465–472. doi:10.1084/jem.184.2.465
Bouche C, Quail DF. Fueling the tumor microenvironment with cancer-associated adipocytes. Cancer Res. 2023;83(8):1170–1172. doi:10.1158/0008-5472.CAN-23-0505
Budnik B, Amirkhani H, Forouzanfar MH, Afshin A. Novel proteomics-based plasma test for early detection of multiple cancers in the general population. BMJ Oncol. 2024;3(1):e000073. doi:10.1136/bmjonc-2023-000073
Chanier T, Chames P. Nanobody engineering: Toward next-generation immunotherapies and immunoimaging of cancer. Antibodies (Basel). 2019;8(1):13. doi:10.3390/antib8010013
Chandarlapaty S, Sawai A, Scaltriti M, et al. AKT inhibition relieves feedback suppression of receptor tyrosine kinase expression and activity. Cancer Cell. 2011;19(1):58–71.
Deng Z, Wu S, Wang Y, Shi D. Circulating tumor cell isolation for cancer diagnosis and prognosis. EBioMedicine. 2022;83:104237. doi:10.1016/j.ebiom.2022.104237
Gonzalez DM, Medici D. Signaling mechanisms of the epithelial–mesenchymal transition. Sci Signal. 2014;7(344):re8. doi:10.1126/scisignal.2005189
Guo A, Villén J, Kornhauser J, et al. Signaling networks assembled by oncogenic EGFR and c-Met. Proc Natl Acad Sci USA. 2008;105(2):692–697.
Han L, Zhang JS, Zhou J, et al. Single VHH-directed BCMA CAR-T cells cause remission of relapsed/refractory multiple myeloma. Leukemia. 2021;35(10):3002–3006. doi:10.1038/s41375-021-01269-3
Hanahan D. Hallmarks of cancer: New dimensions. Cancer Discov. 2022;12(1):31–46. doi:10.1158/2159-8290.CD-21-1059
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57–70. doi:10.1016/S0092-8674(00)81683-9
Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144(5):646–674. doi:10.1016/j.cell.2011.02.013
Huang H, Wu T, Shi H, et al. Modular design of nanobody–drug conjugates for targeted delivery of platinum anticancer drugs with an MRI contrast agent. Chem Commun (Camb). 2019;55(35):5175–5178. doi:10.1039/c9cc01391a
Irving M, Lanitis E, Migliorini D, et al. Choosing the right tool for genetic engineering: Clinical lessons from chimeric antigen receptor T cells. Hum Gene Ther. 2021;32(19–20):1044–1058. doi:10.1089/hum.2021.173
Liang SB, Fu LW. Application of single-cell technology in cancer research. Biotechnol Adv. 2017;35(4):443–449. doi:10.1016/j.biotechadv.2017.04.001
Liu Y, Wang H. Biomarkers and targeted therapy for cancer stem cells. Trends Pharmacol Sci. 2024;45(1):56–66. doi:10.1016/j.tips.2023.11.006
Long Y, Mao C, Liu S, et al. Epigenetic modifications in obesity-associated diseases. MedComm (2020). 2024;5(2):e496. doi:10.1002/mco2.496
Lorentzen CL, Haanen JB, Met Ö, Svane IM. Clinical advances and ongoing trials on mRNA vaccines for cancer treatment. Lancet Oncol. 2022;23(10):e450–e458. doi:10.1016/S1470-2045(22)00372-2
Maiuri AR, O’Hagan HM. Interplay between inflammation and epigenetic changes in cancer. Prog Mol Biol Transl Sci. 2016;144:69–117. doi:10.1016/bs.pmbts.2016.09.002
Ma X, Wang M, Ying T, Wu Y. Reforming solid tumor treatment: The emerging potential of smaller format antibody–drug conjugates. Antib Ther. 2024;7(2):114–122. doi:10.1093/abt/tbae005
Mao Y, Wang X, Zheng F, et al. The tumor-inhibitory effectiveness of a novel anti-Trop2 Fab conjugate in pancreatic cancer. Oncotarget. 2016;7(17):24810–24823. doi:10.18632/oncotarget.8529
McKinney, S. M., Sieniek, M., Godbole, V., Godwin, J., Antropova, N., Ashrafian, H., … & Shetty, S. (2020). International evaluation of an AI system for breast cancer screening. Nature, 577(7788), 89-94.
Qi C, Liu C, Peng Z et al.
Claudin-18 isoform 2-specific CAR T-cell therapy (satri-cel) versus treatment of physician’s choice for previously treated advanced gastric or gastro-oesophageal junction cancer (CT041-ST-01): a randomised, open-label, phase 2 trial
The Lancet, 2025; 405, 2049-2060
Ren SC, Qu M, Sun YH. Investigating intratumour heterogeneity by single-cell sequencing. Asian J Androl. 2013;15(6):729–734. doi:10.1038/aja.2013.106
Rikova K, Guo A, Zeng Q, et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell. 2007;131(6):1190–1203.
Somasundaram K. BRCA1 and BRCA1 Genes and Inherited Breast and/or Ovarian Cancer: Benefits of Genetic Testing. Indian J Surg Oncol. 2010 Sep;1(3):245-9. doi: 10.1007/s13193-011-0049-7. Epub 2011 Mar 11. PMID: 22693372; PMCID: PMC3244237.
Su Z, Xiao D, Xie F, et al. Antibody–drug conjugates: Recent advances in linker chemistry. Acta Pharm Sin B. 2021;11(12):3889–3907. doi:10.1016/j.apsb.2021.03.042
Ubaid A, Khan Z, Ualiyeva D, et al. Chimeric antigen receptor T cell structure, its manufacturing, and related toxicities: A comprehensive review. Adv Cancer Biol Metastasis. 2022;4. doi:10.1016/j.adcanc.2022.100035
Woo V, Alenghat T. Epigenetic regulation by gut microbiota. Gut Microbes. 2022;14(1):2022407. doi:10.1080/19490976.2021.2022407
Woitok M, Klose D, Niesen J, et al. The efficient elimination of solid tumor cells by EGFR-specific and HER2-specific scFv-SNAP fusion proteins conjugated to benzylguanine-modified auristatin F. Cancer Lett. 2016;381(2):323–330. doi:10.1016/j.canlet.2016.08.003
World Health Organization. Global cancer burden growing amidst mounting need for services. 2024. https://www.who.int/news/item/01-02-2024-global-cancer-burden-growing–amidst-mounting-need-for-services
Xia W, Mullin RJ, Keith BR, et al. Anti-tumor activity of GW572016: A dual tyrosine kinase inhibitor blocks EGF activation of EGFR/ErbB2 and downstream Erk1/2 and AKT pathways. Oncogene. 2002;21(41):6255–6263.
Xu G, Liu W, Wang Y, et al. CMG901, a Claudin18.2-specific antibody–drug conjugate, for the treatment of solid tumors. Cell Rep Med. 2024;5(9):101710. doi:10.1016/j.xcrm.2024.101710
Yaremenko AV, Khan MM, Zhen X, et al. Clinical advances of mRNA vaccines for cancer immunotherapy. Med. 2025;6(1):100562. doi:10.1016/j.medj.2024.11.015