
"I have used a wide variety of secondaries and Jackson ImmunoResearch has consistently been the best. The fluorophores are bright and stable and their selective (x reactivity removed) secondaries have always shown species specificity in multiple labeling."
Janet Duerr, Ohio UniversityRating: 5.0
For researchers who perform Western blotting following immunoprecipitation, antibodies specific for light chains or Fc fragments allow unobstructed detection of antigens in the 50 kDa or 25 kDa ranges, respectively.
When labeled secondary antibodies specific for both heavy and light chains of IgG, e.g. anti-IgG (H+L), are used to detect protein bands on Western blots following immunoprecipitation (IP), two heavy bands appear (Figure 1 A) corresponding to the heavy (50 kDa) and light chains (25 kDa) of the precipitated primary antibody. These bands usually obscure detection of any protein of interest with a molecular weight near 50 kDa or 25 kDa. However, when labeled anti-IgG, Light Chain Specific antibodies are used for detection, they bind only to the light chain band on the blot (Figure 1 B) and to light chains on the native primary antibodies used for detection. Therefore, a 50 kDa protein may be detected on blots without interference from the heavy chain of the precipitating IgG.
Light chain specific antibodies are available directed against goat, mouse, rabbit, rat and sheep. They have been adsorbed to minimize cross-reactivity with immunoglobulins from many other species, which also may be present on blots.
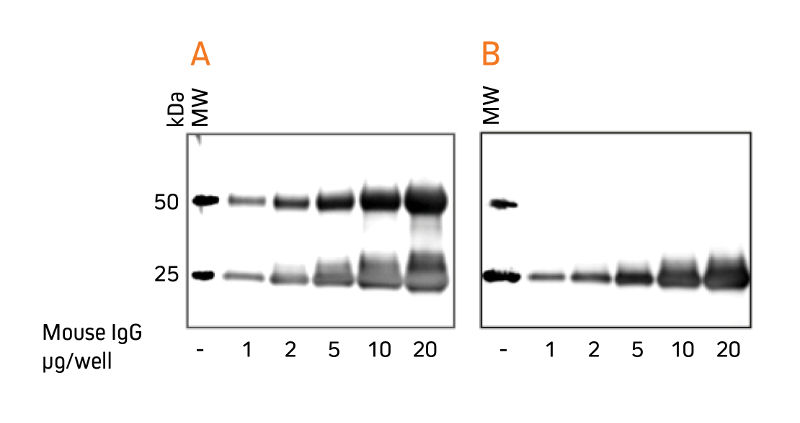
Gels were loaded with reduced and denatured Mouse IgG, whole molecule.
After SDS-PAGE and transfer to nitrocellulose, blots were blocked with BSA (10% w/v). After incubation with secondary antibody, blots were developed with ECL substrate. Blots were imaged simultaneously, with auto exposure time based on bright bands.
A: The gel was probed using HRP-conjugated Goat Anti-Mouse IgG (H+L) (115‑035‑003), revealing bands corresponding to both heavy chains (50 kDa) and light chains (25 kDa).
B: The gel was probed using HRP-conjugated Goat Anti-Mouse IgG, light chain specific (115‑035‑174), revealing only the 25 kDa band corresponding to Ig light chains. The IP antibody heavy chain is not detected, allowing visualization of the protein of interest near 50 kDa.