
"I have used a wide variety of secondaries and Jackson ImmunoResearch has consistently been the best. The fluorophores are bright and stable and their selective (x reactivity removed) secondaries have always shown species specificity in multiple labeling."
Janet Duerr, Ohio UniversityRating: 5.0
Background staining may be observed if a labeled secondary antibody is not adsorbed to minimize recognition of endogenous tissue Ig. When a primary antibody is the same species as the tissue under study (e.g. mouse primary used on mouse tissue), blocking endogenous Ig suppresses the off-target signal.
To block endogenous immunoglobulins on cells or tissue sections, incubate with an excess (20-40 μg/ml) of unconjugated Fab antibody just after blocking with 5% normal serum. Blocking efficiency can be confirmed by eliminating the primary antibody from the protocol and incubating with labelled secondary antibody. It may be necessary to increase the concentration of Fab antibody up to 100 μg/ml to suppress signal from high levels of endogenous IgG.
To avoid displacement of the Fab antibody by the labelled secondary antibody, a light post-fixation with glutaraldehyde may be necessary, provided that it does not affect antigenicity of the target proteins. Fab antibodies are not as effective for blocking immunoglobulins in Western blotting or ELISA applications. For more information see our blog article: A guide to selecting control and blocking reagents.

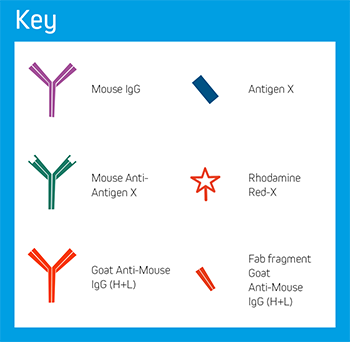

Step 1. Samples may express endogenous immunoglobulins, in this example mouse IgG.
Step 2. After blocking with normal serum, incubate with an excess of unconjugated Fab antibody, in this example Fab fragment Goat Anti-Mouse IgG (H+L). Wash.
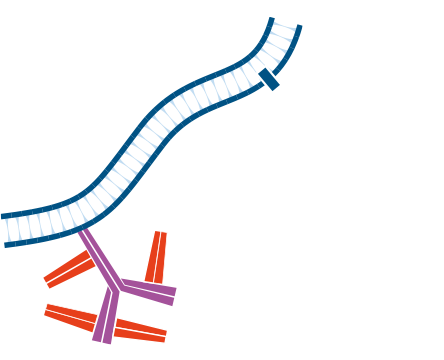
Step 3. Incubate with primary antibody, in this example Mouse Anti-Antigen X. Wash.
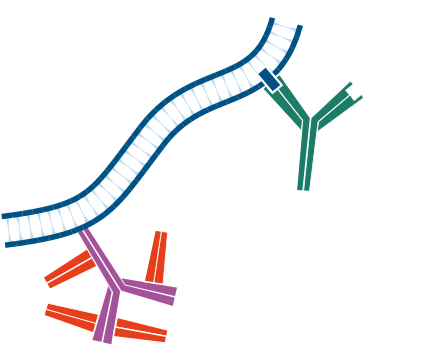
Step 4. Incubate with conjugated secondary antibody, in this example Rhodamine Red™‑X-Goat Anti-Mouse IgG (H+L). Wash.

Monovalent Fab Fragment Affinity-Purified Antibodies for Blocking and Double Labeling Primary Antibodies from the Same Host Species
View Protocols
FabuLight antibodies are Fab fragment secondary antibodies specific to the Fc region of IgG or IgM primary antibodies.
Read More