"I have used a wide variety of secondaries and Jackson ImmunoResearch has consistently been the best. The fluorophores are bright and stable and their selective (x reactivity removed) secondaries have always shown species specificity in multiple labeling."
Janet Duerr, Ohio UniversityRating: 5.0
Immunogold labeling or cytochemistry, is a technique used in Electron Microscope imaging and diagnostic devices such as lateral flow tests. A primary antibody for the protein of interest is detected by a secondary antibody conjugated to colloidal gold nanoparticles. Colloidal gold nanoparticles have a range of applications, when conjugated to antibodies these include high-resolution imaging and diagnostic uses. Different sizes of nanoparticle are available to cater for the technique employed.

Gold nanoparticles produce a phenomenon called surface plasma resonance (SPR) upon their interaction with light, leading to a colored output from the particle which is detectable by eye. The color ranges from bright red through to blue and infrared as the size of the particle increases. This property is exploited in their application to lateral flow immunoassays where a readout requires a colored output that is visible without special equipment.
The electron-dense nature of colloidal gold nanoparticles makes them ideal “marker” molecules for electron microscopy, and double labeling can be successfully performed by combining two sizes of particle, their sizes distinguishable under the microscope allowing differentiation of two target proteins without the need for fluorescence microscopy.
ImmunoGold colloidal gold reagents are available in a range of particle sizes allowing the appropriate size to be chosen for brightfield or electron microscopy, and lateral flow immunoassays.
| Control Reagents | Product Code |
|---|---|
| 40 nm Gold ChromPure Chicken IgY (IgG), whole molecule | 003-400-003 |
| 40 nm Gold ChromPure Goat IgG, whole molecule | 005-400-003 |
| 40 nm Gold ChromPure Human IgG, whole molecule | 009-400-003 |
| 40 nm Gold ChromPure Human IgM (myeloma), whole molecule | 009-400-012 |
| 40 nm Gold ChromPure Human Serum IgA, whole molecule | 009-400-011 |
| 40 nm Gold ChromPure Mouse IgG, whole molecule | 015-400-003 |
| 40 nm Gold ChromPure Rabbit IgG, whole molecule | 011-400-003 |
| Streptavidin | Product Code |
| 40 nm Gold Streptavidin | 016-400-084 |
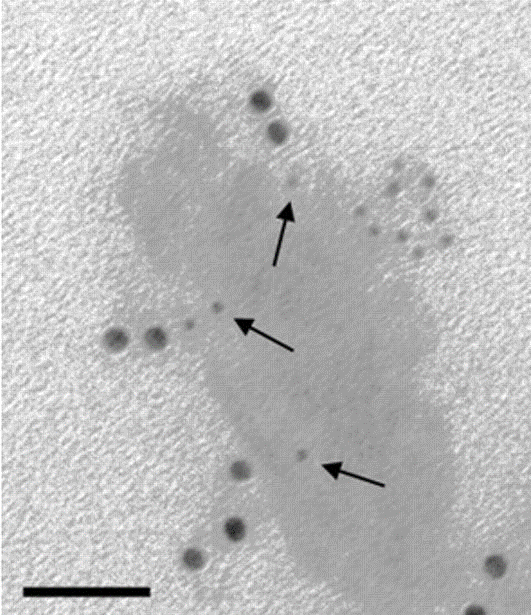
EM Grade Immunogold nanoparticles are distinguished from other commercial preparations by careful separation of monomeric particles from small aggregates using ultracentrifugation in density gradients. The resulting monomeric colloidal gold-protein complexes are recommended for multiple labeling applications, as different antigenic sites can be distinguished by particle size. The complexes are suspended in sterile-filtered buffer containing stabilizers and a preservative.
For staining applications, ImmunoGold reagents offer excellent tissue penetration due to their small particle size (Dixon et al., 2015). For transmission (TEM) and scanning electron microscopy (SEM) EM Grade 6, 12 and 18 nm may be appropriate. The 4 nm size may be used for electron microscopy in studies that require smaller particles since they are relatively uniform in size (coefficient of variation less than or equal to 15%), though small aggregates are not removed from this grade. The 4 nm particles are not suitable for multiple labeling with EM Grade reagents, since size uniformity is paramount and aggregated material may be mistaken for a larger particle.
Lateral flow Immunoassays (LFIAs) require a conjugate that generates a signal which can be read out by eye, but also requires physical properties that enable them to be used in single read formats. Each binding event should produce the strongest signal possible.
40nm colloidal gold conjugates are popular for LFIAs because they produce intense color. This makes readout easy, combined with the simplicity of their conjugation they offer a consistent and reliable reporter molecule.
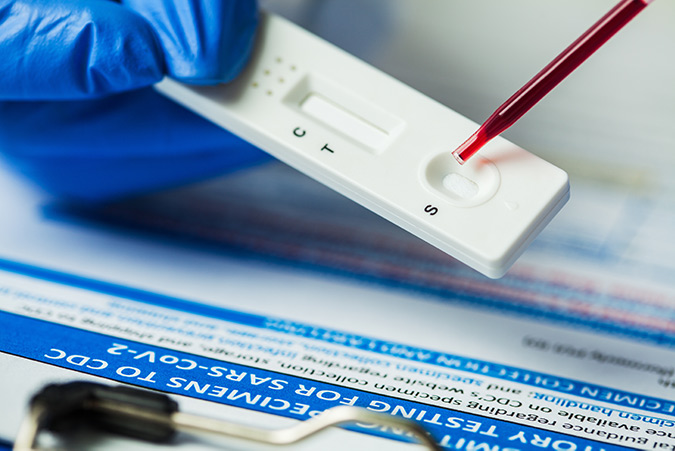
Color intensity is due to the plasmon absorbance and light scattering characteristics of the gold’s electron shell (Huang & El-Sayed, 2010), and the particles’ ability to pack at high density on the test and control lines,due to their small size.
Antibodies are commonly conjugated with gold particles passively via electrostatic and hydrophobic interactions. The two entities are mixed in a low ionic strength buffer, followed by blocking with polyols or proteins like albumin or casein.
In addition to the antibodies listed here, we can also conjugate 40nm gold particles to other diagnostically relevant antibodies we produce, contact us to discuss your requirements.
As well as secondary antibodies, we provide Chrompure™ purified proteins conjugated to 40nm gold as control reagents.
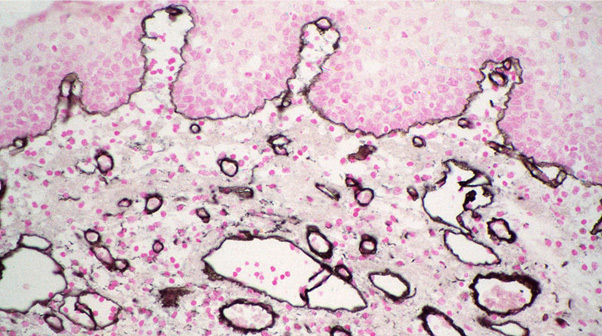
Silver enhancement allows the excellent penetration properties of ImmunoGold reagents to be used with light microscopy. The gold particles act as a nucleation site for the silver ions, which accumulate around the particle until enough contrast is generated to be visualized (Dixon et al., 2015). A detailed protocol for silver enhancement, using reagents that are easily prepared in the laboratory, is provided with all orders for LM Grade products. Alternatively, silver enhancement kits are commercially available.
Signal intensity is relatively independent of particle size when silver enhancement is used, so all particle sizes may be used for light microscopy or immunoblotting. For light microscopy, 4 nm particles (LM Grade) may penetrate tissues better than larger particles.
All LM Grade colloidal gold-protein complexes are freeze-dried in buffer with stabilizers and a preservative. After reconstitution, they may be frozen in aliquots for extended storage.