"I have used a wide variety of secondaries and Jackson ImmunoResearch has consistently been the best. The fluorophores are bright and stable and their selective (x reactivity removed) secondaries have always shown species specificity in multiple labeling."
Janet Duerr, Ohio UniversityRating: 5.0
Imaging with VHH antibodies (Single-domain antibodies, NANOBODIES®) is in its infancy, and their use in super-resolution microscopy has seen promising results (Pleiner et al., 2015). NANOBODY® primary antibodies can be used directly to achieve high-resolution images. However, with a poor range of conjugates available and the potential of paratope compromise during chemical labeling, the use of conjugated epitope tag-directed secondary reagents may be required (Traenkle and Rothbauer, 2017). Here we demonstrate immunofluorescence microscopy of GFP expression in HEK 293 cells using Anti-GFP NANOBODIES® and conjugated Anti-Alpaca VHH domain Antibodies developed by Jackson ImmunoResearch (JIR) in place of tag-directed detection.
Confocal Immunofluorescence using NANOBODIES® is a developing field. VHH antibodies or single-domain antibodies derived from the variable domains of camelid heavy chain-only antibodies offer novel binding characteristics and the opportunity to achieve higher resolution imaging.
VHH antibodies produced recombinantly are between 12 and 15 kDa, making them less than a 10th the size of whole immunoglobulins (Ig) or a 3rd of the size of Fab Fragments. Better sample penetration due to their small size, combined with high solubility and favorable binding affinity, makes NANOBODIES® attractive reagents for studying protein-protein interactions (Begheins and Getteman, 2017).
NANOBODIES® can be used directly or in combination with a conjugated secondary antibody. When used directly, NANOBODIES® are often linked to chemical compounds, such as fluorophores, metal chelates, or radionuclide tracers. However, chemical conjugation can negatively impact the physical properties of the NANOBODY®, such as ligand binding affinity and solubility. Regioselective conjugation via amino acid tags, such as added cysteines, may reduce conjugate impact but could have alternate negative effects such as reduced expression in recombinant systems or reduced protein solubility (Pleiner et al., 2015).
Until recently, detection was directed against purification or detection tags, such as poly-HIS or Myc tags, depending on how the NANOBODY® was produced (de Bruin et al., 2016; Peyrassol et al., 2016) or enzyme tags (Traenkle and Rothbauer, 2017). Detection via epitope tags is not always possible due to their effect on expression and localization, and the availability of detection reagents specific to them. Anti-Alpaca IgG, VHH domain antibodies facilitate immunofluorescence techniques using NANOBODIES®, with detection directed against the VHH domain, not an epitope tag.
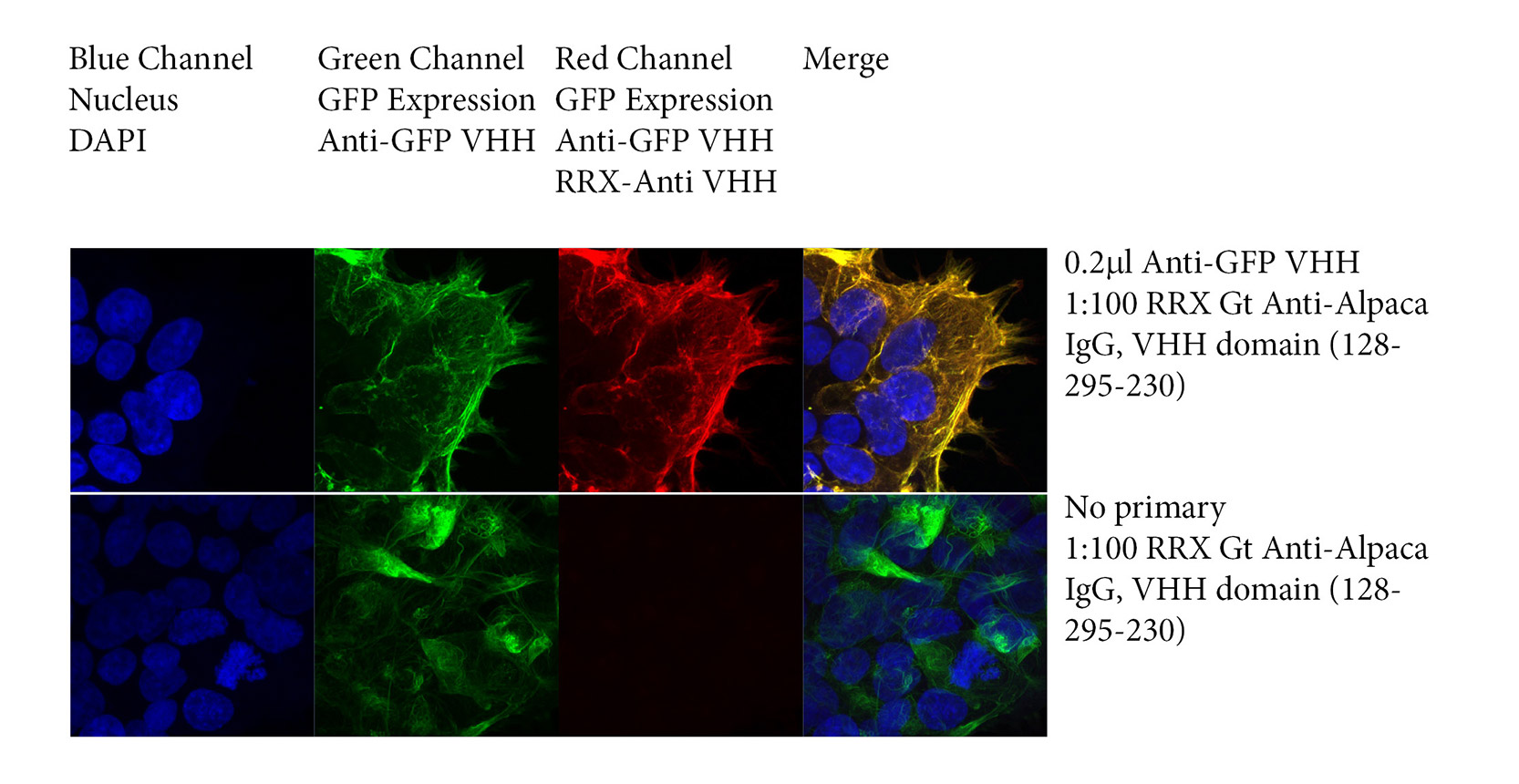
Figure 1 demonstrates immunofluorescence with VHH primary antibodies using JIR’s secondary antibodies directed against Alpaca IgG, VHH domain. Briefly, HEK293T cells were transfected with Green Fluorescent Protein (GFP) and probed with a recombinant Anti-GFP NANOBODY®, followed by JIR’s Rhodamine Red™‑conjugated Goat Anti-Alpaca, VHH domain (128‑295‑230) whole IgG antibody. Nuclei were stained with DAPI.

HEK293T cells were cultured in DMEM medium with 10% FBS at 37 °C with 5% CO2. At approximately 30% confluence, cells were seeded into Lab-Tek II chambered coverglass for 24 h before fixation.
Slides were washed 3x in PBS and fixed in a mixture of 3% paraformaldehyde and 0.1% glutaraldehyde in PBS; washed 3x with PBS; permeabilized with 0.25% (v/v) Triton X-100 in PBS;
Slides were washed 3x with PBS; blocked with 5% (w/v) normal goat serum (005‑000‑121).
Slides were incubated with primary antibody at 0.2μl/ml; washed 3x with PBS; stained with the secondary antibody at 1:100 or 1:500; washed 3x with PBS; post-fixed in a mixture of 3% paraformaldehyde and 0.1% glutaraldehyde in PBS; washed 3x with PBS.
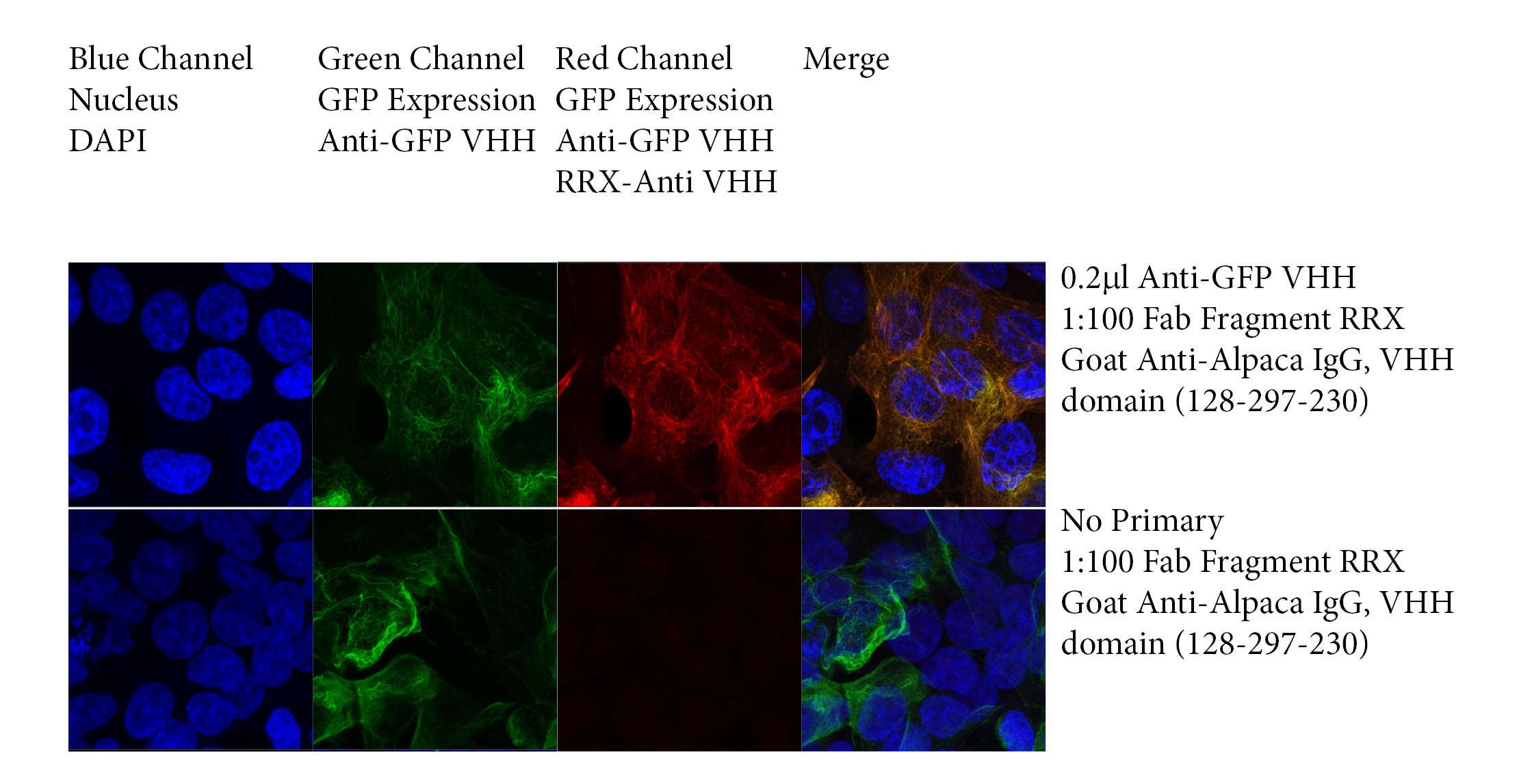
GFP expression was collected in the Green channel, GFP expression was detected by Anti-GFP and Anti-VHH antibody in the Red channel. DAPI staining of nuclei was collected in the Blue channel. Slides were imaged using a Zeiss 880 confocal microscope.
NANOBODIES® are promising reagents for super-resolution microscopy. Being small, the VHH antibody decreases its conjugates’ distance from the target protein, reducing the point spread function and allowing greater resolutions to be achieved. A popular application of super-resolution microscopy is to identify protein-protein interactions, using “Bait and prey” experiments. A fluorescent protein is targeted by a fluor-conjugated NANOBODY®, and the subsequent co-localization, FRET (Forster-resonance-energy-transfer) or LRET (Lanthanide-based resonance energy transfer), confirms protein interaction. (Künzl et al., 2017).
JIR’s 50 kDa conjugated Fab Fragment Anti-Alpaca IgG, VHH domain antibodies can be used in combination with a primary VHH antibody to provide a binding complex much smaller than traditional antibody-antibody pairs. In this regard, Fab fragments may be favorable partners for super-resolution microscopy (Carrington et al., 2019), and may provide greater reporter molecule flexibility than available with directly conjugated NANOBODIES®.
The following experiment shows the detection of GFP in transfected HEK 293T cells with a recombinant Anti-GFP NANOBODY® in combination with JIR’s monovalent Rhodamine Red™‑X- Fab Fragment Goat Anti-Alpaca IgG, VHH domain antibody (128‑297‑230).