
"I have used a wide variety of secondaries and Jackson ImmunoResearch has consistently been the best. The fluorophores are bright and stable and their selective (x reactivity removed) secondaries have always shown species specificity in multiple labeling."
Janet Duerr, Ohio UniversityRating: 5.0
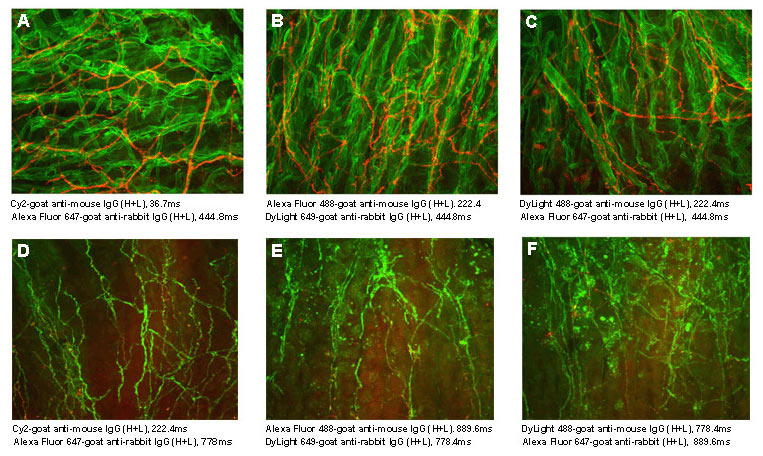
Among currently available fluorescent dyes, the cyanine dyes are better able to withstand the harsh dehydration and embedding conditions required for mounting sections in non-polar plastic media, such as DPX and Permount™. The cyanine dyes are brighter in the non-polar environment than in an aqueous medium, resulting in less acquisition time in the confocal microscope than that required for DyLight and Alexa Fluor® dyes (Figure 1), even though those dyes are brighter in aqueous mounting media.
The major advantages of plastic over aqueous mounting media are brightness, contrast, and longevity of the fluorescence. Cy2, Cy3, and Cy5 are available conjugated to a selection of secondary antibodies for multiple labeling, streptavidin, and purified IgG controls. For multiple labeling, see our listings of Cy2, Cy3, and Cy5 conjugates. Figure 2 illustrates the discrete spectra that provide effective separation for multiple labeling. For single labeling protocols that do not require highly cross-adsorbed antibodies, choose Cy3-conjugated antibodies from the lists of whole IgG affinity-purified secondary antibodies. Note that Cy3 conjugates are bright and photostable in the aqueous environment as well as in non-polar media. This is in contrast to Cy2 and Cy5, which are outperformed in aqueous media by Alexa Fluor® conjugates.
Use the spectra viewer below to compare Cyanine with other fluorophores, along with instrument specifics (laser/filter) for suitability in your assay.
| Fluorophore | Excitation Peak (nm) | Emission Peak (nm) |
|---|---|---|
| Cyanine, Cy2 | 492 | 510 |
| Indocarbocyanine, Cy3 | 550 | 570 |
| Indodicarbocyanine, Cy5 | 650 | 670 |
Table 1. Approximate peak wavelengths of excitation and emission for Cyanine fluorophores conjugated to affinity-purified antibodies. Only approximate values are given for purposes of comparing one fluorophore with another. Actual values may vary depending on the spectrofluorometer used in each laboratory.